Pioneering new Code of Practice on stem cell-based embryo models in research
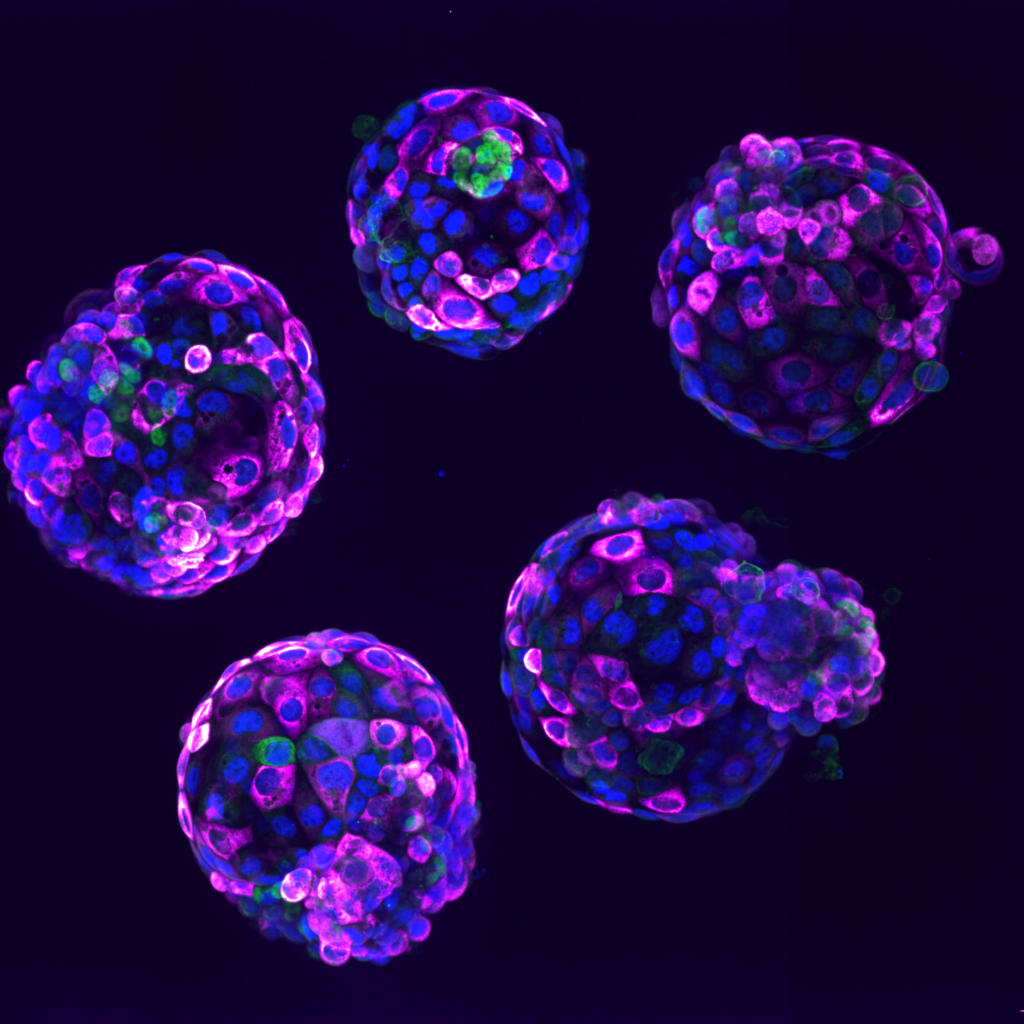
3. Human stem cell-based embryo model – Blastoids. Blue marks all nuclei, the green label marks cells of the inner cell mass, and the pink label is a readout of a ribosomal protein. Credit Irene Zorzan and Teresa Rayon, Babraham Institute.
The first ever UK guidelines for the generation and use of stem cell-based embryo models in research have been created by a partnership that includes University of Exeter researchers.
The guidelines have been developed by a working group of experts from a range of institutions across the UK, representing world-leading expertise in sciences, law, ethics and regulation. The group was led the University of Cambridge, in partnership with the Progress Educational Trust.
Stem cell-based embryo models (SCBEMs) are three-dimensional biological structures that mimic aspects of early human embryo development, which raises various ethical considerations. They can be created in the lab from stem cells, and can provide new insights into critical stages of early human development that are normally inaccessible to researchers. Research using embryo models can advance understanding of early human development and possible causes of infertility.
The use of stem cell-based embryo models is not regulated explicitly by existing UK law, which has created uncertainty for scientists and held back research. The new SCBEM Code of Practice proposes clear guidance and oversight processes, to bring greater transparency and openness to the research and reassurance to the public.
Professor Austin Smith, of the University of Exeter, who contributed to the Code of Practice alongside Exeter colleague Dr Ge Geo, said: “The ability to mimic the earliest stages of human embryo development using stem cells opens important new ways to study causes of human infertility and miscarriage. The UK is a world leader in this research. Ethical oversight is essential for public confidence and the new Code of Practice provides a transparent framework that will be welcomed by scientists in the field.”
The previous lack of a regulatory framework for the use of SCBEMs in research has left scientists and research organisations uncertain about the acceptable boundaries of their work, both legally and ethically, and unsupported in their decision-making. Signing up to the SCBEM Code of Practice will remove these obstacles. The guidelines, released today, provide the first code of practice for the use of stem cell-based embryo models in the UK.
“The new Code of Practice provides processes for decision-making in research using stem cell-based embryo models so that scientists can proceed confidently, while maintaining public trust in this vital area of research,” said Kathy Niakan, Chair of Cambridge Reproduction and Professor of Reproductive Physiology at the University of Cambridge, and member of the SCBEM Code of Practice Working Group.
Key points of the Code
The new SCBEM Code of Practice sets out standards to make sure that research using SCBEMs is rigorous, upholds ethical principles and maximises the potential benefits. Key to this will be a dedicated Oversight Committee that will review each proposed research project.
The Code recognises that there must be a limit to how long embryo models can be grown in the lab. However, many different types of embryo model are being developed, representing distinct developmental stages and developing at different rates, making it impossible to impose a single fixed limit. Instead, researchers are asked to provide clear justification of the length of their experiments on a case-by-case basis.
The Code prohibits any human SCBEM from being transferred into the womb of a human or animal, or being allowed to develop into a viable organism in the lab.
“Embryo models have huge potential and we want to realise this, while also limiting the risks. The new Code of Practice will allow stem cell-based embryo models to be grown in the lab long enough to gain meaningful biological understanding, but the Code asks researchers to fully justify what they’re doing in scientific and ethical terms,” said Professor Niakan.
The SCBEM Code of Practice Working Group was made up of researchers and practitioners at institutions across the UK, including experts in science, law, ethics and regulation. In drawing up the Code, the team consulted widely with researchers, practitioners, and major funders and regulators of embryo model research, both within the UK and internationally.
The SCBEM Code of Practice Working Group included researchers and practitioners from the Babraham Institute, Biolawgy, Brighton and Sussex Medical School, the Centre of Medical Law and Ethics at King’s College London, the Francis Crick Institute, Hull York Medical School, the Medical Research Council, Newcastle University, University College London, the University of Cambridge, the University of Edinburgh, the University of Exeter and the University of Manchester. The work was also informed by a public dialogue, held earlier this year, to explore public attitudes towards research involving embryo models.
The Code is not legislative, but the teamproposes that it should be adopted by UK researchers, funders, research organisations, professional societies and publishers.
Widespread use is expected to set a new precedent for reporting research utilising stem cell-based embryo models and deter the funding and publication of research that fails to meet the standards set out in the Code. As a result, adherence to the Code will become an essential part of credible science, providing increased transparency and accountability for embryo model research.
Given the rapid pace of research using embryo models, the Code will be reviewed regularly.
The work was funded by the BBSRC, the University of Cambridge Impact and Knowledge Exchange fund, and UKRI Sciencewise.
To access the Code, visit: www.repro.cam.ac.uk/scbemcode



